The mode of action of [emergency contraception, EC] has become he subject of heated debate in North America and in several Latin American and Caribbean countries. The main question is centred on whether or not EC prevents pregnancy by interfering with post-fertilization events. This issue is of importance for many people who consider that a new human life begins at the time that fertilization is completed. Accordingly, interference with post-fertilization events would lead to loss of human life. In spite of a lack of scientific evidence to support a post-fertilization effect, this possibility is used as an argument to turn legal, political and religious constituencies against the availability and use of EC. (Ortiz, et al, 2004)
In order to satisfy my own curiosity and my critics, I’ve reviewed recent scientific literature related to the question of whether or or not Plan B is abortifacient. I do not wish to appear to be in any way “rooting” for Plan B and/or its supporters. I am not. However, I have been very frustrated by the way that many of my fellow pro-lifers have repeatedly stated confidently that Plan B sometimes acts after fertilization and is therefore abortifacient. I do not believe such confidence is supported by scientific evidence. The goal of this literature review is to present a fair appraisal of the likelihood of postfertilization effects caused by Plan B to the pro-life community.
For the most part my analysis will proceed in chronological order, but I’ll begin with a newer article (Croxatto, Ortiz, and Müller, 2001) that provides an brief primer on the relevant reproduction science.
[Hormonal emergency contraception, HEC] prevents pregnancy, in which case it acts after fertilization.The fertile days of the menstrual cycle are the days in which an act of sexual intercourse can give rise to pregnancy.These are the 5 days that precede ovulation and the day of ovulation [4]. Thus, spermatozoa have to wait one to 5 days in the female genital tract before encountering the ovum in most cases. This interval provides an opportunity to interfere with the migration and function of spermatozoa and/or with the process of ovulation. EC pills may prevent the encounter of spermatozoa with the ovum; and, even if the two gametes do come in contact, fertilization may not proceed to completion.
The efficiency of fertilization in human beings is very low in comparison with most mammals. Under ideal circumstances,when intercourse takes place during the most fertile days, the chance that fertilization will take place does not exceed 50% [5] and it is plausible that even minor alterations in the preceding processes will greatly decrease this probability. It has been shown that HEC pills do interfere with pre-fertilization events [6]. When the pills are taken too late to prevent fertilization there are two possible outcomes: (a) that HEC will not be effective and the method fails; (b) that when a woman uses HEC, she does not know whether she takes the pills before or after ovulation, before or after fertilization. For ethical and logistic reasons, it has not been possible to segregate groups of women who take EC after fertilization so as to assess its effect on the establishment of pregnancy. Hence, there is no direct evidence, either for or against, the hypothesis that EC pills prevent pregnancy by interference with post-fertilization events.
There is no direct evidence against the hypothesis that EC pills prevent pregnancy by interference with post-fertilization events. However, as we shall see, there is much indirect evidence against it. Furthermore, since a negative cannot be proven, the burden of proof is on whomever offers a positive hypothesis. However, in order to satisfy critics who claim that the EC Plan B (a.k.a. DL-Norgestral, levonorgestral, LNG) is abortifacient, research has been conducted in order to establish that pre-fertilization effects can account for the effectiveness of the drug.
Some of the discrete steps of the reproductive process whose theoretical interference by EC could prevent pregnancy are:
- follicle maturation
- the ovulatory process
- sperm migration into and through the fallopian tube, including adhesion of spermatozoa to the epithelium needed to acquire and maintain their fertilizing capacity
- fertilization
- zygote development in the fallopian tube
- zygote transport through the fallopian tube
- preimplantation development within the uterus
- uterine retentiveness of the free laying morula or blastocyst
- endometrial receptivity
- blastocyst signaling, adhesion and invasiveness
- corpus luteum sufficiency and responsiveness to hCG
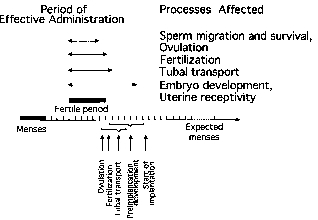
Figure 1 illustrates the chronology of some of these steps within the normal conceptional cycle of women and the time period within which EC needs to act to interfere with each one of them.
(Croxatto, et al., 2001)
A handy reminder of the general order of hormones related to fertility is the acronym FELOP, which stands for
- Follicle Stimulating Hormone (FSH)
- Estrogen (E1, E2, E3 = estrone, estradiol, estriol)
- Luteinizing Hormone (LH)
- Ovulation
- Progesterone (P,P4)
For more detailed treatment of reproduction suitable for lay consumption, I recommend Taking Charge of Your Fertilityby Toni Weschler.
Before looking at the most recent salient research, let’s examine a couple oft-sited peer-reviewed literature reviews. The first, Postfertilization Effect of Hormonal Emergency Contraception (Kahlenborn, Stanford, and Larimore, 2002), is often cited by pro-lifers as presenting evidence that Plan B is abortifacient.
The question as to whether hormonal EC sometimes acts after fertilization to prevent implantation, called a postfertilization effect (i.e., early abortion), is important and could have far-reaching implications given the differing attitudes in regard to its use and related issues such as informed consent, emergency department rape protocols, and conscience clauses. Postfertilization effect refers to any effect that reduces the survival rate of the zygote/embryo after fertilization, usually prior to clinical recognition of pregnancy. We use the term early abortion synonymously with postfertilization effect. We recognize that some physicians,4 geneticists, and ethicists have arbitrarily defined human life as beginning after implantation, thereby eschewing the possibility of an early abortion prior to implantation. However, we recognize the traditional definition of pregnancy: “the gestational process, comprising the growth and development within a woman of a new individual from conception through embryonic and fetal period to birth,†where conception is defined as “the beginning of pregnancy, usually taken to be the instant that a spermatozoon enters an ovum and forms a viable zygote.â€5
A someone who believes that life begins at conception, I’m in agreement with this motivational introduction. However, its obvious bias and polemical tone does make me a little uncomfortable from an objective scientific viewpoint. Moving on:
In a previous review6 of the mechanism of action of [oral contraceptives, OCs], we concluded that they act at times via a postfertilization effect, that is, after fertilization and prior to the clinical recognition of pregnancy. However, the Yuzpe regimen and Plan B have a different dose and time course for use, which may result in different actions on the reproductive system. This article reviews data on the use of the Yuzpe regimen and Plan B with regard to their mechanisms of action and any potential ethical implications of those mechanisms.
That’s as far as you’re going to read from that paper on this blog. The authors consistently refuse to disambiguate the Yuzpe regimen (a mix of ethinyl estradiol – an estrogenic hormone – and levonorgestrel – a synthetic progestogen) and Plan B (levonorgestrel alone). Consequently, since both are forms of EC, any evidence that Yuzpe might be abortifacient is implied to damn Plan B as well. Their earlier literature review of OCs that they cite suffers from a similar “guilt by association” approach. Since the effects of LNG are not presented clearly, separate from those of the Yuzpe regimen, this review only serves to add confusion and misunderstanding to the current discussion. Were it not for its frequent citation by various members of the pro-life movement, I would not mention it at all. On the other hand, the end of the article includes some good points about informed consent that are worth reading. However, though interesting, they are not directly relevant to this review.
Let’s look at another literature review, Mechanism of action of hormonal preparations used for emergency contraception: a review of the literature (Croxatto, et al., 2001), that clearly and distinctly presents the results of LNG studies separate from other EC methods. Though undertaken by proponents of EC, the review does not shy away from suggesting that LNG might have postfertilization effects.
Numerous studies have shown that pregnancy can be prevented in a variety of mammalian species by post-coital administration of sex steroid hormones, their synthetic agonistic and antagonistic analogs as well as non-steroidal drugs that share in part their pharmacologic properties. The species that comprise this group, which are most commonly used in the laboratory, e.g., rabbit, rat, mouse, hamster, and guinea pig, differ from the human and other primates in many aspects of their reproductive process. One of them which is crucial for studying the mode of action of postcoital contraception, is the fact that at variance with the human, coitus always precedes ovulation by 12 h or less. This means that coitus either comes after the ovulatory stimulus has taken place (spontaneous ovulators) or induces the gonadotropin surge immediately thereafter (reflex ovulators), leaving little or no chance for post-coital treatments to interfere with pre-fertilization events. Only a few examples, which illustrate what sex steroids do in these species when used in a manner comparable to that of EC, were arbitrarily selected.
[…]
The effect of levonorgestrel administered in the periovulatory period in non-human primates has not been reported. Since its binding affinity to progesterone receptor (PR) and its progestomimetic activity are several-fold higher than P itself [61], one can expect it will share many of the effects of the natural progestin. The effects of P upon fertility, when administered in the periovulatory phase, have not been reported in the monkey. However, exogenous progesterone antagonizes estrogen-induced gonadotropin release required for ovulation [59,62] and causes asynchronous development of glands and stroma in the endometrium [47,63,64]. Progesterone plays a pivotal role in periovulatory and luteal events through receptor-mediated pathways [65– 68]. Thus, a reduction of P bioavailability by inhibition of its synthesis or by competition with its receptor might interfere with ovulation, fertilization, luteal function, and subsequent endometrial development.
These admissions regarding the downsides of relying on animal studies in these matters are important to bear in mind when reading the remainder of their review, as well as recent papers that I will review in subsequent installments. Some of those who regard Plan B as abortifacient refuse to accept the results of such animal studies. However, endochrinologists have little choice in the matters given the bioethical complications of performing studies on humans. Another paper in this review (Ortiz, et. al, 2004) explains.
For ethical and logistic reasons, it has not been possible to segregate groups of women who take EC after fertilization in order to assess its effect on the establishment of pregnancy. Hence, there is no direct evidence that post-coital treatment with LNG prevents pregnancy by interfering with post-fertilization events. On the other hand, animal experimentation allows the investigator to segregate groups treated before or after critical events, such as ovulation, fertilization and implantation, in order to define the contribution of interference with pre- and post-fertilization events to the contraceptive efficacy of the drug. Although extrapolation of the results to humans has limitations, experiments in animals often shed light on possible mechanisms operating in humans.
Animal experimentation provides an important necessary ethical, if imperfect, means for assessing the methods of action for EC. In fact, some of the authors of the Croxatto, et al., proceeded to study the effects of LNG on rats and new-world monkeys.
The following review excerpts represent research that had been completed prior to submission in October, 2000.
There are few studies designed to look at the mechanism of action of LNG in EC and its exact mode of action remains unknown. Moggia et al. [88] proposed that the post-coital contraceptive effect of LNG is due to changes in the endometrium that prevent implantation. Kesseru et al. [89] provided evidence that single administration of LNG 0.4 mg 3 to 10 h post-coitum: a) decreased the number of sperm recovered from the uterine cavity beginning 3 h after treatment; b) caused pronounced alkalization of the intrauterine fluid beginning at 5 h, which immobilized the sperm, and; c) increased the viscosity of the cervical mucus, beginning at 9 h, which denied further passage of sperm to the uterus. Serege´ly [90] suggested that disturbances in LH pulse frequency following LNG administration were involved. Landgren et al. [21] examined the effects of repeated doses of LNG (0.75 mg) given before (days 2, 4, 6, and 8), during (days 9, 11–13, 15, 16, and 19) or after ovulation (days 16, 18, 20, and 22). Administration in the early follicular phase increased the duration of the follicular phase. Treatment around ovulation resulted in varying effects ranging from anovulation or deficient luteal function in some women to normal ovarian function in others. The administration of LNG during the luteal phase was not followed by changes in cycle length or endometrial morphology. Spona et al. [91] administered single or multiple doses of 0.4 mg LNG before or after the LH peak to 6 subjects. Treatment prior to the LH peak suppressed the gonadotropin surge, reduced the levels of E2 and P, and markedly lowered the cervical mucus score, whereas treatment after the LH peak did not alter these parameters. The effects of these multiple administrations cannot be freely extrapolated to the current EC regimen.
Wang et al. [92] compared the effects of 0.75 mg LNG given twice, 12 h apart, when the first dose was administered on day LH-2 versus LH+2. The main endpoints were timing and incidence of ovulation and the status of the endometrium at the time of implantation (LH+7). Preovulatory administration had no effect on ovulation, whereas at the level of the endometrium, it caused divergent effects depending on the time of drug intake. Factors believed to be critical for implantation, such as integrins, steroid receptors, or leukemia inhibitory factor, among others, were changed in ways which are likely to alter endometrial receptivity.
The only study that provides a large enough data base to examine the relationship between coitus-treatment interval and outcome shows that LNG as well as the Yuzpe regimen exhibit an inverse relationship between contraceptive efficacy and the length of time from intercourse to treatment. Pregnancy rates increased from 0.5% when treatment was given within the first 12 h period after intercourse to 4.1% when it was given within the fifth 12 h period (61–72 h) [93]. While this fact does not allow for discriminating between possible modes of action, it does lend support to a significant role of pre-fertilization mechanisms in their contraceptive effectiveness, albeit not necessarily the same ones for both methods. Our hypothesis is that…in the case of LNG the earlier it is given, the better the chances it will interfere with sperm migration and function at all levels of the genital tract.
The proposed mechanisms of action can be briefly summarized as follows.
- 1974: changes in the endometrium that prevent implantation
- 1974: hostility to spermatozoa
- 1975: suppressed gonaotropic surge, reduced levels of E2 and P, thickening of cervical mucus (treatment prior to LH)
- 1982: interference with LH pulse (and therefore ovulation)
- 1989: increased follicular phase duration, anovulation, deficient luteal function (depending on time of treatment)
- 1998: changes to endometrium that could alter receptivity
- 1999: interference with sperm migration and function (treatment after ovulation)
Numerous attempts to determine the involvement of selected steps of the reproductive process in the mechanism by which EC prevents pregnancy have been done. In spite of that, a wide gap of information persists that hinders a clear-cut answer to the question.
With few exceptions, the fact that an entity or a process is altered by the treatment does not necessarily mean that it explains how pregnancy is prevented in real life situations. In this respect, ovulation inhibition can explain by itself how pregnancy is prevented whereas abnormal expression of a given molecule in the endometrium lacks that strength until it is shown that its normal expression is essential for pregnancy to occur. [Emphasis mine]
IOW, no egg means no fertilization, i.e., no baby. On the other hand, changes to the endometrium do not inherently indicate any particular method of pregnancy prevention. A change is only relevant in this context if it is demonstrated to interfere with postfertilization events. This is an important point because it weakens the claims of those who believe that any change to the endometrium strongly suggests that LNG interferes with implantation and is therefore abortifacient.
It is now well recognized that one of the complexities that researchers have to deal with to find a thorough answer is that the mechanism may differ for the same EC treatment depending upon when it is given relative to time of intercourse and also relative to time of ovulation. A single act of intercourse that takes place up to 5 days before ovulation may result in pregnancy in the human. Therefore, many women who request EC receive the treatment before ovulation and possibly before fertilization if ovulation has occurred. Neither the minimum length of time from coitus to fertilization, when the oocyte is waiting for the sperm, nor the shortest interval from ovulation to fertilization, when the sperm is waiting for the oocyte, have been determined in the human. Therefore, the exact theoretical amplitude of the window for acting before fertilization is undetermined, less so the actual window in real cases.
This problem is highlighted in an interesting 2004 paper by Mikolajczyk and Stanford (yes, that Stanford), which describes problems in common methods for predicting fecundity and presents a new method that seems to be less biased. I’ll cover this in some detail in a later installment.
The contraceptive effectiveness of LNG and the Yuzpe regimen has been shown to depend on the intercourse-treatment interval (the easy one to obtain), whereas there is no data for the ovulation-treatment interval (the difficult one to obtain). Given that in 15–25% of the cycles treated with EC, the expected pregnancy is not prevented, chances are that there is a specific window in the cycle in which treatment is more likely to fail. Attempts to pinpoint the stage of the menstrual cycle at which treatment is given to women requesting EC for subsequent correlation with the contraceptive outcome may shed some light on the mode of action of a particular method. Admittedly, even at a research center, it is difficult to get informed consent for such a study given the anxiety that surrounds every case. Since the intercourse-treatment and ovulation-treatment intervals are inter-related, should information about both become available, complex analyses will be needed to estimate how each one relates to the contraceptive outcome.
According to Mikolajczyk and Stanford, the estimated failure rate of 15-25% is based on flawed estimations of fecundity and may be well below the mark. If so, the “residual” pregnancy-preventing capacity that so many insist must be accounted for by postfertilization effects may be illusory.
Most mechanistic studies have attempted to assess to what extent ovulation inhibition is involved. However, none has used ultrasound to confirm follicular rupture and to pinpoint at what stage of follicular development treatment was given. It is clear that EC appears to prevent ovulation in many cases but not so clear what the conditions are in terms of timing of treatment relative to the stage of follicular development. The criteria used to time treatment lacks this precision in practically all studies reviewed. Because ultrasound has not been used, the occurrence of ovulatory dysfunctions, such as luteinized unruptured follicle, has not been determined.
Later studies on new-world monkeys (Ortiz, et al., 2004), surgically sterilized women (Durand, et al., 2001), and women made sterile by tubal ligation or nonhormonal IUDs (Müller, et al., 2003) employed ultrasound to observe follicular development.
Both logistic and ethical constraints prevent designing and performing experiments that can directly address what in fact happens to the crucial biologic entities – sperm, oocyte, zygote or preimplantation embryo – in the genital tract of women who receive EC in comparison to those who receive placebo. The fate of spermatozoa and of the oocyte can be studied without risking the occurrence of conception if either one is absent from the genital tract. It is easy to avoid the presence of sperm for this purpose, without altering the biologic environment. In order to suppress the presence of the oocyte, one could inhibit ovulation using a GnRH-antagonist and give appropriate sex steroid replacement therapy to provide a “normal environment for spermâ€. The effect of EC treatment on sperm could then be studied at centers where retrieval of spermatozoa from the site of fertilization is feasible. In fact, with the exception of Kesseru et al. [89], no other study has focused on the effects of EC upon spermatozoa.
A later literature review (Gemzell-Danielsson and Marions, 2004), which will be discussed in another installment, refers to studies that observed the effects of LNG on sperm migration and function.
Alterations in embryo transport through the fallopian tube or uterus following EC, are also difficult to explore. Delayed transport or retention in the tube cannot be excluded a priori, although no increased incidence of tubal pregnancy has hitherto been reported with the current methods.
Reports, or lack thereof, of increased rates of tubal pregnancy will not be discussed in my review. However, if any of my readers have compelling evidence in this matter, they are encouraged to share them in comments. Tubal pregnancy (a dangerous condition that requires abortion) certainly qualifies as an anti-implantation effect; I have only omitted discussion on this topic due to time constraints as a busy grad student who has his own research to worry about.
Accelerated transport through the tube appears unlikely since neither estradiol nor progesterone given in high doses right after ovulation have this effect in women [102]. Expulsion of the egg from the uterus could result from myometrial effects of wide steroid oscillations.
The first statement means that high doses of estradiol (which is present in the Yuzpe regimen) or progesterone (which is present in both Yuzpe and Plan B) have not induced accelerated transport of a zygote from a fallopian tube. Timing of zygote transport to the uterus is important in the implantation process. Accelerated transport might result in a zygote encountering an inhospitable environment for implantation and therefore be aborted.
The second statement is very confusing. As a nonexpert I found it difficult to parse and am open to correction of the assessment that follows. As I understand it, means that by flooding the body with LNG, some uterine or endometrial effect may be triggered that would evict the egg somehow. That is, a woman using LNG might still ovulate, but that egg might never have a chance to fertilize and any observed changes to the endometrium that might impede implantation in other situations actually contributed to evicting the egg.
Several studies have focused attention on alterations of the endocrine profile during the luteal phase. Luteal insufficiency, caused by EC, cannot be claimed to contribute to pregnancy prevention until it is shown to persist through a hCG challenge test.
Under normal circumstances, following the release of the egg from the ovary, the follicle that held the egg collapses on itself. What remains is called the corpus luteum (Latin for “yellow body”). It remains in the interior ovarian wall and secretes progesterone for 12 to 16 days. At this time in the cycle progesterone is important for preventing the release of additional eggs and causing the thickening of the uterine lining (endometrium). If the latter does not occur, an embryo might not be able to implant.
If I understand the argument being made correctly, observations of luteal insufficiency in studies may simply indicate lack of an embryo. Since hCG (human chorionic gonadotropin) is secreted by an embryo and helps to maintain the corpus luteum, if a bolus of hCG were administered after ovulation and progesterone levels did not rise, then one could conclude that EC had impaired the corpus luteum to the point of making the endometrium inhospitable to implantation. A positive response to the test, however, would indicate that corpus luteum function is not impaired. Such a hCG challenge test would be difficult to perform, however, due to complicated timing issues and bioethical complications regarding performing tests in the possible presence of an embryo.
The most difficult parameter to assess with certainty is endometrial receptivity. Endometrial markers of receptivity have been established so far with certainty only in rodents. Even if endometrial receptivity is shown to be altered by EC, other steps that precede implantation may also be altered enough to interrupt the process at an earlier stage.
IOW, even if the endometrium were rendered entirely hostile to implantation, it would not matter if fertilization had already been prevented via anovulation, impeded sperm migration and/or function, or other means. That is, the endometrial changes may be part of the effect rather than the cause. Remember, correlation doesn’t require causation.
Well, that’s the state of LNG activity research as of late 2000. As it stands at this point, it seems that LNG acts primarily via prefertilization effects. However, there is insufficient evidence to rule out the possibility that it acts after fertilization in some cases. Especially troubling are changes to the endometrium that might affect receptivity. In the next installment, I’ll cover the paper On the mechanisms of action of short-term levonorgestrel administration in emergency contraception (Durand, et al., 2001), which reports a study of the effects of LNG on the pituitary-ovarian axis, corpus luteum function, and endometrium.
References
- Croxatto HB, Devoto L, Durand M, Ezcurra E, Larrea F, Nagle C, Ortiz ME, Vantman D, Vega M, von Hertzen H. Mechanism of action of hormonal preparations used for emergency contraception: a review of the literature. Contraception 2001;63(3):111-121.
- Durand M, del Carmen Cravioto M, Raymond EG, Duran-Sanchez O, De la Luz Cruz-Hinojosa M, Castell-Rodriguez A, Schiavon R, Larrea F. On the mechanisms of action of short-term levonorgestrel administration in emergency contraception. Contraception 2001;64(4):227-234.
- Kahlenborn C, Stanford JB, Larimore WL. Postfertilization effect of hormonal emergency contraception. Ann Pharmacother 2002;36(3):465-470.
- Müller AL, Llados CM, Croxatto HB. Postcoital treatment with levonorgestrel does not disrupt postfertilization events in the rat. Contraception 2003;67(5):415-419.
- Gemzell-Danielsson K, Marions L. Mechanisms of action of mifepristone and levonorgestrel when used for emergency contraception. Hum Reprod Update 2004;10(4):341-348.
- Ortiz ME, Ortiz RE, Fuentes MA, Parraguez VH, Croxatto HB. Post-coital administration of levonorgestrel does not interfere with post-fertilization events in the new-world monkey Cebus apella. Hum Reprod 2004;19(6):1352-1356.
Pingback: just another day of Catholic pondering
Pingback: Lux Venit
Incredible. You’ve done an excellent job, better than I think I could have done.
Thank you. I look forward to more.
What a knotty problem. Thanks for trying to look into.
To those viewing this post on September 19:
For the duration of the day, all of the posts on this blog will appear to have been written by a pirate. Everything will go back to normal at the end of the day.
An excellent detail oriented review of the available research. This should be a particularly useful resource for all those on the internet who would like to understand the meaning and appropriate application of these studies.
Might I suggest meta tags, etc. that would help folks looking for the layman’s read on these studies to find this?
Pingback: Plan B: Literature Review (Part II) @ Ales Rarus
Pingback: Ross Douthat (September 07, 2007) - The Morning After (Culture Wars)